Manufacturers looking to optimise access for their orphan drugs need to navigate a complex environment – this article outlines some of the key drivers and nuances that manufacturers may need to consider.
It is common knowledge that Orphan Drug Designation (ODD) may provide a regulatory head start, but it does not materially shift evidence, pricing, or reimbursement criteria nor is a substitute for demonstrated value in national HTAs. Indeed, published studies across seven European countries and over multiple assessment periods, indicate that approximately onethird of orphan drugs assessed do not receive public reimbursement.1
ODD Overview
ODD is a status granted to medicines in Europe by the European Commission (EC) (and by the MHRA in the UK), intended to treat serious conditions affecting fewer than 5 in 10,000 people.2 Its purpose is to incentivise development and improve access to treatments for rare diseases that otherwise be commercially unattractive.
In the EU, ODD is granted before marketing authorisation (MA), while in the UK it is applied for alongside MA3 (Figure 1). In both cases, ODD provides 10 years of market exclusivity and fee reductions4, with additional protocol assistance available in the EU.2
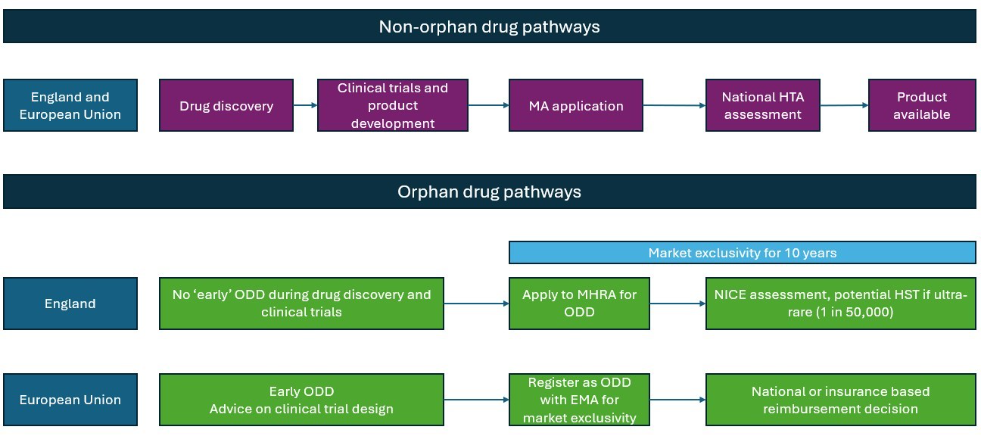
Despite ODD status, reimbursement decision-drivers vary across EU
ODD is granted at the EU level, while reimbursement is decided nationally. At this level, each EU country has different incentive mechanisms and level of methodological flexibility to assess rare and ultra-rare therapies – independent of regulatory ODD status.
Table 1: Impact of ODD on the reimbursement pathways within EU4 and the UK
| Market | How reimbursement pathways consider disease rarity? |
|---|---|
| UK3 | NICE has a highly specialised technology (HST) program, allowing treatments for ultra rare diseases (affecting 1 in 50,000) to benefit from cost effectiveness thresholds of up to £300,000 per QALY. |
| France5 | Full reimbursement under the long-term illness provision (ADL31) Faster HTA timeline: typically reduced from 90 to 30 days |
| Germany6 | Under AMNOG, orphan drugs undergo a partial assessment and are granted unquantifiable benefit, providing sales do not exceed a revenue threshold of €30 million (when full assessment applies)9 |
| Italy7 | Fast-track pricing and reimbursement evaluation timeline of 100 days The submission immediately after a positive CHMP opinion, so it does not require waiting until the EU MA is granted |
| Spain8 | No dedicated orphan route |
Rarity does not equal value
It is important to note that regardless of national level methodological nuances, rarity and unmet need does not change how payers at the national level assess the therapy’s value, therefore a compelling payer value proposition supported by robust evidence remains the most important driver of patient access and price potential. From 2028 onwards, there will be even higher pressure for a robust value proposition with all orphan drugs falling within the scope of the EU Joint Clinical Assessment (JCA), harmonizing clinical assessment and reducing flexibility for divergent national evaluations.10
Degree of acceptable uncertainty is limited
National HTA bodies may accept less mature evidence packages for ODs owing to the practical challenges of conducting randomised controlled trials (RCTs) in low-prevalence diseases; however, depending on the HTA body, products may still need to demonstrate comparative value to justify their cost.
While RCTs remain the gold standard for generating evidence, they are often infeasible for orphan indications because there is no established standard of care. In such cases, alternative approaches such as external control arms (ECAs) may be used to demonstrate incremental benefit. ECAs draw on data from patients outside the clinical trial, including electronic health records and disease or treatment registries, to serve as comparators for single-arm studies. The high uncertainty around ECAs was illustrated by axicel (Yescarta), which initially missed NHS commissioning due to a weak alignment with the SCHOLAR-1 study.11 However, it was still on the Cancer drugs fund, which can be attributed to the product addressing an unmet need. Rare diseases may lack an established standard of care, to ensure successful HTA submissions, control arms should still include the best supportive care (whether this be off-label or symptomatic only), suitable, high quality data sources and a rigorous methodology to minimise risk of bias.12
Low prevalence does not mean affordability
ODs often come with very high per-patient costs, in order to make it commercially viable given the small target populations.13 This small patient population could mean a small budget impact, but high prices can still be the driving factor in an orphan drug not being nationally reimbursed, as was the case for cerliponase alfa where NICE concluded its use would not be an acceptable use of NHS resources based on the products cost-effectiveness estimates.14 This shows the potential for a patient with a rare disease to be less likely to receive treatment than a patient with a more common disease, and therefore to incur lower costs. An analysis looked at the cost of orphan drugs compared to their non-orphan comparator in the EU4 and UK. They found that orphan drugs cost 3 to nearly 7 times as much as non-orphan drugs.15
Beyond initial pricing rationale, market exclusivity can sustain high prices over time by limiting direct competition. While exclusivity was designed to make rare disease R&D investable, manufacturers should not assume that a lack of an established standard of care alone will secure reimbursement at high per-patient prices. In practice, even if a single OD does not translate into a significant budget impact, several ODs reimbursed over time can cumulatively threaten the sustainability of healthcare budgets.
So, what next?
From 2028, the EU Joint Clinical Assessment will further harmonise expectations for robust, mature evidence, making early payer engagement essential for securing funding and timely patient access. Orphan status may shape pathways and timelines, but it rarely substitutes for robust evidence to reduce uncertainty. Given the budgetary pressures payers face in Europe, OD prices will increasingly be scrutinised to align with the level of incremental value they provide to patients, as well as ODs’ combined impact on healthcare systems – rather than compensating or incentivising manufacturers for investing in underserved rare indications
References
- Stawowczyk E, Malinowski KP, Kawalec P, Bobiński R, Siwiec J, Panteli D, Eckhardt H, Simoens S, Agusti A, Dooms M, Pilc A. Reimbursement status and recommendations related to orphan drugs in European countries. Front Pharmacol. 2019;10:1279. doi:10.3389/fphar.2019.01279
- European Commission. Orphan medicinal products. European Commission website. Accessed January 28, 2026. https://health.ec.europa.eu/medicinal-products/orphan-medicinal-products_en
- UK Parliament Parliamentary Office of Science and Technology. Treating rare diseases: the challenge of orphan drugs. POSTnote. Parliamentary Office of Science and Technology website. Accessed January 28, 2026. https://post.parliament.uk/treating-rare-diseases-the-challenge-of-orphan-drugs/
- UK Government. Orphan medicinal products in Great Britain. GOV.UK website. Accessed January 28, 2026. https://www.gov.uk/guidance/orphan-medicinal-products-in-great-britain
- Staatz Business. White paper: orphan drugs. Published July 16, 2020. Accessed January 28, 2026. https://www.staatz.biz/wp-content/uploads/White-paper-orphan-drugs_16072020_FINAL-1.pdf
- Institute for Quality and Efficiency in Health Care (IQWiG). Orphan drugs. Press release. IQWiG website. Accessed January 28, 2026. https://www.iqwig.de/en/presse/press-releases/press-releases-detailpage_58496.html
- Agenzia Italiana del Farmaco. Farmaci orfani. AIFA website. Accessed January 28, 2026. https://www.aifa.gov.it/en/farmaci-orfani
- Badia X, Gil A, Poveda-Andrés JL, Shepherd J, Tort M. Analysing criteria for price and reimbursement of orphan drugs in Spain. Farm Hosp. 2019;43(4):1-15. doi:10.7399/fh.11147
- Simon-Kucher & Partners. Germany: how the GKV-FinStG law is transforming the pharma pricing and reimbursement landscape. Simon-Kucher website. Accessed January 28, 2026. https://www.simon-kucher.com/en/insights/germany-how-gkv-finstg-law-transforming-pharma-pr-landscape
- Mtech Access. EU JCA: Joint Clinical Assessment. Mtech Access website. Accessed January 28, 2026. https://mtechaccess.co.uk/eu-jca/
- National Institute for Health and Care Excellence. Committee papers — axicabtagene ciloleucel for treating diffuse large B-cell lymphoma and primary mediastinal large B-cell lymphoma (TA872). NICE website. Published January 26, 2023. Accessed February 18, 2026. https://www.nice.org.uk/guidance/ta872/documents/committee-papers-3
- Mangla KK, Kolovos S, Lisica A, et al. Acceptability of external control-arm use in nononcology health technology assessment submissions. J Comp Eff Res. 2025;15(1):e250073. Published 2025 Dec 12. Accessed [insert access date]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12711071CMS Law-Now. Orphan drugs and reasons behind their exorbitant prices. CMS website. Published February 2020. Accessed January 28, 2026. https://cms-lawnow.com/en/ealerts/2020/02/orphan-drugs-and-reasons-behind-their-exorbitant-prices
- Althobaiti H, Seoane-Vazquez E, Brown LM, Fleming ML, Rodriguez-Monguio R. External control arms in oncology drug development: methodological and regulatory considerations. Clin Pharmacol Ther. 2023;113(3):515-525. https://pmc.ncbi.nlm.nih.gov/articles/PMC9957503/
- National Institute for Health and Care Excellence. Final draft guidance – Cerliponase alfa for treating neuronal ceroid lipofuscinosis type 2 (review of HST12). NICE website. Issue date: August 2025. Accessed February 18, 2026. https://www.nice.org.uk/guidance/hst34/documents/674
- Perez A, Gas N, Rives V, Parlato G, Gaddam K, Vialard L. Assessing the influence of the orphan drug status on medicine prices in the EU4 and UK. ISPOR Europe 2022 presentation; Vienna, Austria; 2022. Poster presentation. https://www.ispor.org/docs/default-source/euro2022/ispor-2022—assessing-the-influence-of-the-orphan-drug-status-on-medicine-prices-in-the-eu4-uk-pdf.pdf?sfvrsn=679e6926_0. Accessed February 18, 2026