Introduction
In the EU novel biologics generally need to undergo a national Health Technology Assessment (HTA) to secure reimbursement, in order for their clinical and economic value to be assessed. Biosimilars offer clinical comparability to the originator biologic at a lower cost, and as such can often circumvent formal HTA submissions. This article will address where a formal HTA is and is not needed. Pricing topics, such as pricing rules and international reference pricing (IRP), or how to demonstrate the value of a biosimilar, will not be covered here but can be found in other Remap articles from ‘Biosimilar Month’.
HTA requirements for biosimilars
Where is no HTA required?
In the EU5, there are three countries where no HTA is needed for a biosimilar launch: Germany, Spain and the UK. In these cases, the regulatory assessment of bioequivalence to the originator is considered sufficient, and only price and other matters relating to the funding and procurement of the product act as barriers to patient access.
In Germany, a biosimilar is available once a price has been registered with the Lauer Taxe, and patients can usually access the product following tenders in the retail market or direct contracting in the hospital setting. In Spain, only a price proposal is needed for biosimilar launch, with limits on the list price, following which the product is available for procurement. In the UK, a product can be procured once price is set with the Department of Health and Social Care, although in a hospital setting tendering is often utilised.1
Where may HTAs be used?
On the other hand, France and Italy can require a submission for biosimilars, although fast-tracked or otherwise abridged submission are sometimes sufficient.
In France, biosimilars may be evaluated by the Transparency Commission (TC) but an ASMR (improved medical benefit) rating of V will be automatically assigned to the product, with the same reimbursement conditions as the originator. Biosimilars can avoid TC assessment via a fast-track pricing discussion with CEPS (the Economic Committee for Health Products).
The Italian Medicines Agency (AIFA) will carry out ‘standard’ pricing and reimbursement negotiations for biosimilars unless the proposed price is considered an ‘obvious convenience’, a status which is not formally defined but expected to be above the mandated 20% discount to the originator. In this case, AIFA places the biosimilar in the same reimbursement class as its originator. As seen in many cases, tendering for biosimilars is common in both countries, although only for hospital biosimilars in France.1
Looking beyond the EU5
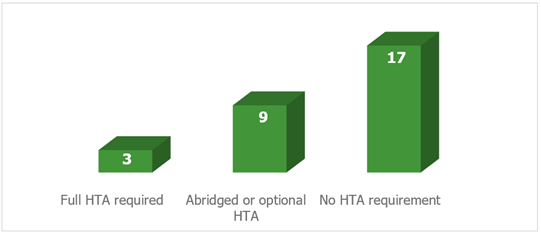
Looking further than the EU5, a review we carried out of requirements across twenty-nine European countries found only three that required a full HTA submission for biosimilars: Estonia, Slovenia and Sweden. Figure 1 shows the breakdown of requirements.
An exception to the rule?
While the above section lays out that there is no need for HTA submission in the UK, this is not to say that no biosimilars have ever been assessed by the National Institute for Health and Care Excellence (NICE). In 2021 a Multiple Technology Assessment (MTA) was conducted for the use of adalimumab, etanercept, infliximab, certolizumab pegol, golimumab, tocilizumab and abatacept in the treatment of rheumatoid arthritis.
Biosimilars for adalimumab, etanercept and infliximab were available, and the introduction of adalimumab and etanercept biosimilars was the reason for this MTA partially reviewing a previous appraisal. The cost and access implications (e.g., regional variation) of the biosimilars were considered in the MTA.
However, a key point here is that while the MTA could have impacted future access and funding for the products at an active ingredient level, the MTA was not a critical step for access at launch for the biosimilars.
Conclusion
To revisit our initial question, it is clearly too simple to say that HTAs have no importance when it comes to biosimilar launch in the EU5. However, importance is far lower than what we may expect for a ‘traditional launch’ – for example that of an originator biologic. If you would like to hear how we can support you in facilitating the submissions and discussions that are needed to launch a biosimilar in the EU5, please get in touch.
Sources:
- Medicines for Europe. MARKET REVIEW – BIOSIMILAR MEDICINE MARKETS: POLICY OVERVIEW. https://www.medicinesforeurope.com/wp-content/uploads/2021/03/Biosimilar%20Market%20Review-Final.pdf Accessed 3rd October 2022.
- NICE. TA715: Adalimumab, etanercept, infliximab and abatacept for treating moderate rheumatoid arthritis after conventional DMARDs have failed. https://www.nice.org.uk/guidance/ta715 Accessed 3rd October 2022.