Introduction
Despite the impact of the COVID-19 pandemic with many resources steered towards management of the virus, pharmaceutical expenditure in oncology treatments for the US is predicted to continue its double-digit growth, estimated to be by 12% in the next two years1.
The rush of innovative oncology treatments, accompanied by the expected continued rising costs of oncology drugs, provides a public health challenge to patients and health systems in the US. An affordable and sustainable market access landscape is key to ensure patient access to life-saving therapeutics. Consequently, companies must be equipped with a robust pricing and market access strategy that meets the requirements of the evolving oncology market and considers patient value alongside affordability.
Financial toxicity and cancer treatments
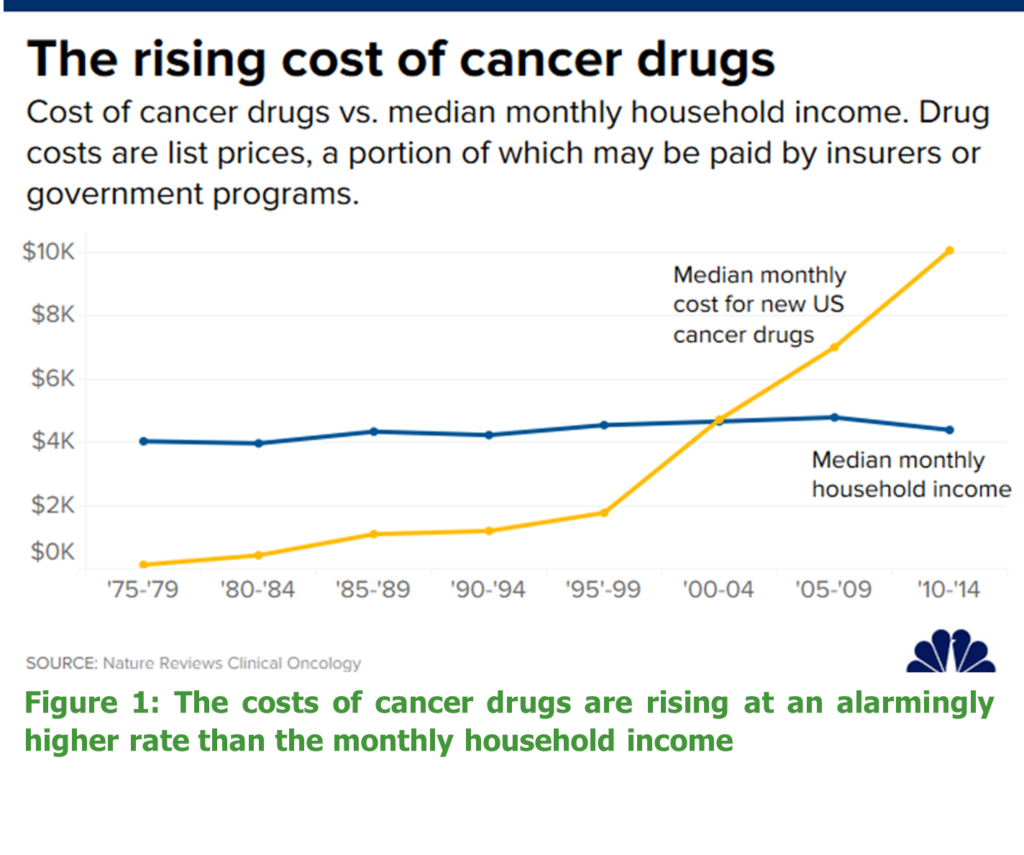
Spending on oncology drugs is increasing faster than spending on other health conditions. In the US, spending on cancer drugs has doubled since 2012, reaching almost $50 billion in 2017, with costs expected to double again by the end of this year2. Furthermore in the US, cancer drug prices for novel treatments remain two to six times higher than in the rest of the world3.Indeed, the cost of cancer drugs is rising at a much higher rate than household income (see Figure 1). As a result, the US oncology market has recently received significant negative press regarding the cost of new oncology treatments – for one, it is reported a cancer patient is more likely to claim bankruptcy than the average person4. Virtual monopolies, high research and development costs and lack of regulation over drug prices has contributed to high costs of cancer therapeutics in the US, where cancer drug costs accounted for more than 50% of the total cost of oncology care in 20185. A cost-analysis conducted on 65 cancer drugs, demonstrated that the median monthly cost at drug launch was $11,755 per patient in the US compared to between $7355 and $8305 per patient in the EU6. The study further demonstrated no association existed between launch price and clinical benefit.
It would be natural to assume that elevated cancer drug prices are due to the greater value for patients these drugs are providing. Unfortunately, this is not always the case. For example, the current seven available PD-(L)1 therapies, which are used for the treatment of non-small-cell lung cancer (NSCLC), on the US market have list prices in the region of $150,000 per year7, but response rates remain below 50%1. Competitive market dynamics are the main driver for drug price points in the US, reflecting what the market can bear rather than an association with clinical benefit. This is largely the result of big pharmaceutical companies remaining unchallenged in the oncology market and having the ability to set prices higher than the level at which the US Health Technology Assessment (HTA) body, the Institute for Clinical and Economic Review (ICER) would consider to be cost-effective.
Correlating patient value with price
There is growing demand in the US to adopt a framework linking drug price to value; manufacturers must consider the improvement in patient-relevant outcomes in their pricing discussions. This environment shift must consider the value of oncology therapeutics from the patient perspective not just from a payer perception. For novel cancer treatments, bodies in the US such as ICER and Sloan Kettering are beginning to consider the economic and clinical benefit, respectively.
In regulatory and payer decision making, the inaccurate correlation between endpoints used in oncology trials and overall survival of cancer patients is commonly critiqued. In the case of PARP inhibitors for the treatment of ovarian cancer, ICER issued an evidence report in 2017 which concluded that the high cost of the drugs did not align with benefit to patients and further research on improvement in overall survival was required before use was recommended8. This demonstrates how utilising frameworks to evaluate relevant patient endpoints can assist in P&MA decision making and prevent oncology drugs that yield little or uncertain benefit from dominating the market.
How high is the ceiling?
Value frameworks in the US have much to offer, but novel oncology treatments provide illustrative examples of where these frameworks are yet to have an impact. For example, the number of Chimeric antigen receptor (CAR)-T therapies on the US market is expected to increase from a total of two in 2019 to 24 by 20241. CAR-T cell therapies are among the most expensive cancer therapies, they set up price points with the promise of great potential but with limited data at launch. In the US, a real-world data study showed that of 74 patients receiving this therapy, almost 40% did not have a durable response5. A further 12% had adverse complications resulting in further healthcare costs of more than $1 million5. Whilst these novel treatments provide growing excitement in oncology pharmaceuticals, current value frameworks need to be improved in order to handle more complex market-access considerations. These include scenarios such as costly one batch mechanisms, and uncertainty over long-term efficacy for cancer therapeutics. This will bring novel, clinically meaningful therapies to patients, providers and payers.
Conclusion
Continued elevating oncology pharmaceutical expenditure raises the question whether the US marketplace can continue to afford high-cost treatments or whether measures will be required to limit the increase. Current price elevation presents challenges for patient access to drugs with clinically meaningful outcomes. Moving forward, companies must consider a pricing and market access strategy that demonstrates the clinical value alongside an affordable drug price.
Improving value- or outcomes-based frameworks could help ensure the availability of higher-value medicines. Sophisticated decision-making tools are required to support market access and deliver effective treatments to patients and physicians. Manufacturers should consider in their future market access strategies greater collaboration with payers, clinicians and patients in the development of new oncology treatments and subsequent pricing discussions; to align on clinical value, comparable outcomes and cost containment. This will be key for pharmaceutical companies to ensure they remain in a dynamic and changing oncology market landscape.
Here at Remap consulting we can harness our vast experience in pricing and market access to guide pharmaceutical companies where they should focus their efforts to ensure their share in the pharmacological oncology market remains. If you think your company could benefit from our expertise, please get in touch.
Sources:
- Albrecht, B et al. Delivering innovation: 2020 oncology market outlook. Mckinsey & Company. Accessed on: March 24, 2022. https://www.mckinsey.com/industries/life-sciences/our-insights/delivering-innovation-2020-oncology-market-outlook
- Spending on Cancer Meds in the U.S. Doubled from 2012-2017 – expected to double again by 2022 to $100 Billion. Newsroom. 2018. Accessed on: March 24, 2022. https://www.iqvia.com/newsroom/2018/05/iqvia-institute-for-human-data-science-study-spending-on-cancer-meds-in-the-us-doubled-from-2012-201
- Leighl, N. et al. An Arm and a Leg: The rising cost of cancer drugs and impact on access. ASCO Education Book. 2021; 41:e1-e12. DOI: 10.1200/EDBK_100028
- Konish, Lorie. Cancer sends many patients into bankruptcy. Advisors are stepping up to stop that. 2019. Accessed on: March 24, 2022. https://www.cnbc.com/2019/11/24/cancer-puts-many-patients-into-bankruptcy-advisors-are-trying-to-help.html
- Wehrwein, P. Have we hit the breaking point with oncology drug prices? Managed Healthcare Executive. 2021; 31(7). Accessed on: February 25, 2022. https://www.managedhealthcareexecutive.com/view/have-we-hit-the-breaking-point-with-oncology-drug-prices-
- Volkinger, K et al. Analysis of launch and post-approval cancer drug pricing, clinical benefit, and policy implications in the US and Europe. JAMA Oncol. 2021; 7(9). Accessed on: March 24, 2022. https://jamanetwork.com/journals/jamaoncology/article-abstract/2781390
- Neil Grubert. FDA committee deals a blow to the prospect of low-priced Chinese oncology drugs in the U.S. Accessed on: March 24, 2022. https://www.linkedin.com/posts/neil-grubert_oncology-china-fda-activity-6898198680143556608-s_9D/
- Final report on PARP inhibitors for Ovarian Cancer. Press Releases. 2017. Accessed on: March 24, 2022. https://icer.org/news-insights/press-releases/ovarian-cancer-parp-final/