Introduction
The early access to medicines scheme (EAMS) was introduced in April 2014 based on a recommendation from the Ministerial Industry Strategy Group for an early access scheme. Its aim was to allow people with life-threatening or seriously debilitating conditions access to medicines before marketing authorisation when there is a clear unmet need. Here, we provide an overview of the EAMS process, our opinions on the impact of the scheme so far, and considerations for manufacturers submitting products through this process.
The PIM / EAMS Process
EAMS criteria and summary of the process
The EAMS process broadly is split into 3 stages: the promising innovative medicine (PIM) designation, the EAMS scientific opinion, and EAMS patient access within the NHS.
ONE: The PIM designation indicates that a product may be eligible for the EAMS based on early clinical data and has the potential to address an unmet need. The company must make an application which includes information on the disease background and unmet need, as well as non-clinical and clinical data on the product. A fee of £3,624 is payable at this stage. If successful, the PIM designation will be issued after an MHRA scientific meeting. NICE recommends that companies which receive a PIM designation also seek joint scientific advice.
TWO: When a positive PIM designation is obtained, companies can then proceed to the EAMS scientific opinion. The company must attend an MHRA pre-submission meeting to ensure that the product is suitable, and then submit an EAMS dossier. NICE’s Office for Market Access also offers companies the opportunity for a supplementary meeting to discuss the data collection plans during the EAMS period. The fee for assessment of the scientific opinion for a new chemical/biological medicinal product is £25,643. To obtain a positive scientific opinion, the product must several criteria:
- The drug must treat a life-threatening/ seriously debilitating condition with a high unmet need
- The product must be likely to offer a significant advantage over current methods in the UK
- The potential adverse effects of the product are outweighed by the benefits
- The applicant can supply and manufacture the product to a consistent quality standard.
Once a positive scientific opinion is communicated, the MHRA will publish a public assessment report (PAR) and the EAMS treatment protocol, defining the key aspects of the product and the scientific opinion. The opinion lasts for a year and can be renewed for a fee of £4,154.
THREE: The EAMS period then starts, in which the company supplies the drug free of charge, collects real-world evidence, and prepares for the NICE technology appraisal. NICE plans drugs with a PIM designation or positive EAMS opinion into its work programme as a priority. If NICE makes a positive recommendation, the product is commissioned within 30 days of NICE guidance (compared to the 90 days allotted for non-EAMS commissioning), and early access will cease. If NICE does not recommend the product, the company must agree to a clear exit strategy with the relevant bodies
Outcomes from the PIM/EAMS process so far
Between April 2014 and April 2022, 119 PIM designations were granted, and 45 positive scientific opinions were awarded, with 78% of PIM applications successfully resulting in a designation, and 75% of EAMS applications resulting in a positive scientific opinion. This contrasts with 605 positive EMA opinions in the same timeframe.
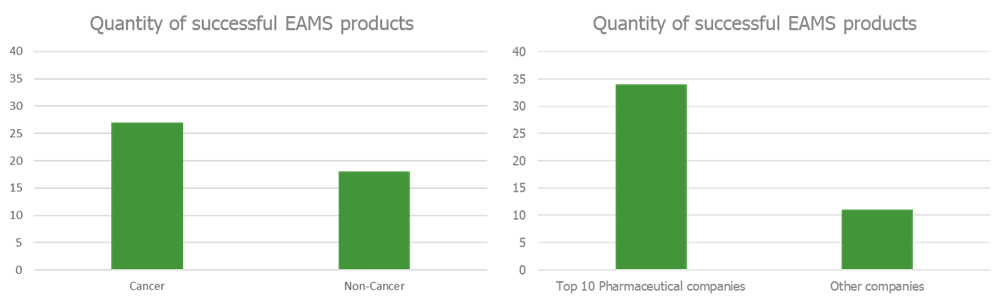
Positive scientific opinions were awarded more often to products with indications for cancer compared to non-cancer conditions (Figure 1a), with most of these being for drugs made by top 10 pharma companies (Figure 1b). 35% of positive scientific opinions were for the checkpoint inhibitors nivolumab, pembrolizumab and atezolizumab combined. The average duration of the EAMS access period is 169 days (around 6 months), however some products have had vastly higher access durations. For example, Santhera Pharmaceuticals’s Raxone had an access period of over 3 years. On average around 6 positive scientific opinions are given each year, in line with the original expectations of the scheme.
Implications on NICE appraisals
The collection of data whilst in the EAMS has contributed in recent NICE appraisals to reducing uncertainty regarding some aspects of the clinical data and health economic modelling. For example, the use of avalglucosidase alfa use in clinical practice through the EAMS supported its long-term efficacy and safety in NICE TA821. The clinical practice experience and data from the time of berotralstat in EAMS suggested its efficacy increased over time, and supported the application of a continuation rule in the model in NICE TA738. EAMS data that supported the generalisability of the patient characteristics in the VISION trial to the NHS helped get a positive recommendation for tepotinib in TA789 in its second meeting. Finally risdiplam, another product originally not recommended by NICE, had EAMS data from 203 patients showing that the product met an unmet need. This aided a positive recommendation at the second meeting.
Considerations for manufacturers
From an industry perspective, the EAMS process has numerous benefits for companies. EAMS offers a valuable opportunity for companies to engage in early dialogue with NICE, the MHRA and other relevant bodies about product uptake within the NHS. A positive EAMS opinion results in a prioritised NICE appraisal and faster implementation of a positive decision, and the real-world evidence collected during the EAMS period can be used during the NICE appraisal. The provision of the drug free-of-charge to the NHS can also reflect well on the manufacturer.
However, there are challenges that manufacturers must also take into consideration. The lack of NHS funding prior to market authorisation may be a significant barrier to entry, especially for smaller companies. The application process itself is also resource intensive, involving submissions, regulation, medical compliance, and testing for safety at different stages of the EAMS process. This extends to the necessity of periodic report submission on exposure and safety data in addition to dosing/preparation errors. Finally, there remains the chance that market authorisation or reimbursement is not obtained, resulting in the need for an exit strategy for the manufacturer.
Conclusion
The evidence suggests that most companies apply for a PIM designation only, and that the EAMS is primarily reserved for larger pharmaceutical companies which can handle the resource requirements, fees and risk associated with the scheme. Another scheme focusing on fast access for innovative medicines is the Innovative Licensing and Access Pathway (ILAP), which was introduced in 2021 with the aim of accelerating the time to market for innovative drugs. ILAP offers benefits including early engagement with key bodies in UK healthcare such as the MHRA, NICE, and the SMC, and offers an HTA access forum tool to help address critical questions. The data so far suggests that the ILAP has been more popular than the PIM, with 160 applications for innovation passports from 2021 to 2023 compared with 152 PIM applications from 2014 to 2022.
Sources:
- Patil, S., 2016. Early access programs: Benefits, challenges, and key considerations for successful implementation. Perspectives in Clinical Research. 7(1), pp.4-8. DOI: 10.4103/2229-3485.173779
- The Early Access to Medicines Scheme: An independent review. National Health Service. https://www.gov.uk/government/publications/independent-review-of-early-access-to-medicines-scheme-eams. Accessed January 25th January 2023.
- PIM designation statistics. MHRA. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1066819/PIM_D_stats_April_2022.csv/preview. Accessed 25th January 2023.
- Apply for the early access to medicines scheme (EAMS). MHRA. https://www.gov.uk/guidance/apply-for-the-early-access-to-medicines-scheme-eams#overview. Accessed 25th January 2023.
- O’Connor, D.J., McDonald, K., Ping Lam, S., 2017. Earlier Patient Access – the UK Early Access to Medicines Scheme (EAMS). The Journal of Medicine Access. DOI: 10.5301/maapoc.0000009
- Note to describe procedures at NICE to support the Early Access to Medicines Scheme. NICE. https://www.nice.org.uk/media/default/about/who-we-are/policies-and-procedures/eams-process-jan-16.pdf. Accessed 25th January 2023.
- Expired early access to medicines scheme scientific opinions. MHRA. https://www.gov.uk/government/publications/early-access-to-medicines-scheme-expired-scientific-opinions/expired-early-access-to-medicines-scheme-scientific-opinions#raxone-to-treat-the-decline-of-respiratory-function-in-patients-with-duchenne-muscular-dystrophy. Accessed 25th January 2023.
- Early Access to Medicines Scheme. ABPI. https://www.abpi.org.uk/media/dfikg1ka/early_access_to_medicines_scheme.pdf. Accessed 25th January 2023.
- Innovative Licensing and Access Pathway. MHRA. https://www.gov.uk/guidance/innovative-licensing-and-access-pathway. Accessed 25th January 2023.
- Boysen, M. Data presented at Utrecht Centre for Pharmaceutical Policy and Regulation Winter Meeting. NICE. 11th January 2023.