The year 2026 marks a significant milestone for the European Union’s (EU) Health Technology Assessment (HTA) reform, as industry stakeholders will gain visibility on the full process, including the decisions and approaches implemented by assessors and co-assessors up to now.
This article highlights key lessons from 2025 and examines how the EU HTA reform is likely to evolve in 2026. We’ll outline practical actions manufacturers can take now to ensure they are fully prepared for the JCA, based on the learnings we now have access to.
What Year One (2025) taught us
The EU’s Health Technology Assessment (HTA) framework, including the Joint Clinical Assessment (JCA) and Joint Scientific Consultation (JSC), entered into force in January 2025. The initial roll-out focused on oncology medicines with a new active substance and Advanced Therapy Medicinal Products (ATMPs) as shown in Figure 1 below.1
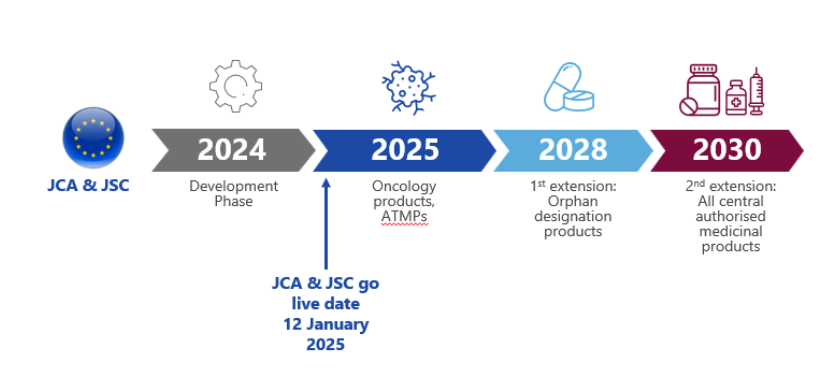
The number of JCAs initiated in 2025 was lower than expected (approximately half of projections, ~25), with only 12 assessments, including 10 for oncology therapies and 2 ATMPs.2 The reason for the lower-than-expected number of submissions may be partly attributed to sponsor quiet deferrals to avoid being in the first wave, while implementation “teething issues” such as fragmented public communication and operational uncertainty played out. The first year of JCA implementation was also characterised by lack of transparency into the rationale behind published decisions, reducing manufacturers’ predictability in dossier planning.
In November 2025, the European Federation of Pharmaceutical Industries and Associations (EFPIA) reflected on the PICO scoping and consolidation process* so far, and three key observations were underlined:
- Development of initial PICO proposals: lacked transparency on how assessors/co-assessors formulate initial PICO proposals.
- PICO consolidation process: allowed significant flexibility, despite established definitions and methodology.
- Relevance of the JCA scope: different standards of care across Europe required multiple PICOs, leading to more complex JCA questions. As a result of attempts to simplify the process, some Member States may find their needs insufficiently reflected. Conversely, oversized dossiers also risk misinterpretation and can reduce the JCA’s usefulness for national decisions.
As a result of these observations, the EFPIA advocates for a refined and clearer PICO scoping process to improve efficiency and ensure national relevance. They recommend routine manufacturer input into draft PICOs, plus ongoing process fixes and timely updates (e.g., with timely communication via established channels such as FAQs) ahead of any formal regulatory revision.4 If the European Commission observes these recommendations, more transparent and closer engagement with manufacturers might be expected in 2026.
*An initial stage of the JCA process is the PICO (Population, Intervention, Comparators, and Outcomes) scoping, where the JCA gathers input from Member State HTA bodies and then consolidates those inputs into a final set of PICO research questions that will guide the rest of the clinical assessment.3
What will change in 2026 and what it means for manufacturers?
In 2026, the HTA Coordination Group (HTACG) estimates a meaningful increase in the number of JCAs, comprising 35 for new active substances in oncology, 15 ATMPs and 5 high-risk medical devices.5 This also represents an increase in the breadth of assessments. As the number and breadth of assessments increases, industry stakeholders will continue to closely monitor how JCA outputs will translate into pricing and reimbursement outcomes.
The EU HTA Regulation will introduce its first JCAs for certain high-risk medical devices, specifically Class IIb and III devices and in vitro diagnostics from class D, while ongoing JCAs for oncology drugs and ATMPs will continue.1 To facilitate this process, the HTACG has published a new set of guidance.8 This will provide useful insight on how medical device assessment might deviate from that of pharmaceuticals, something which not well understood currently.
With the predicted increase in the number of JCAs, capacity issues may linger into 2026. For instance, the European Commission has announced that there will be a maximum of 17 slots for scientific advice (8–12 for medicines, 2–5 for medical devices).7 However, demand is likely to exceed the number of available slots, so manufacturers should be prepared to apply early.
EU HTA 2026 readiness: priorities for market access leaders
As the EU HTA Regulation continues to evolve, the following priorities will be essential to support Market access leaders in ensuring both JCA and national HTA evidence requirements are met:
| Key Activities | Description |
|---|---|
| Leverage learnings from early JCAs | Analyse output from finalised JCAs to understand assessor and co-assessor influence, including how differences in Member State evidence interpretation shape JCA outcomes |
| Early PICO modelling | Conduct initial scenario planning/PICO simulations, and leverage learnings from completed JCAs, to anticipate the level of flexibility and complexity that can be expected |
| Plan Selective Use of Joint Scientific Consultation | Given the limited availability of JSC slots, consider alternative options to inform your evidence strategy, such as payer advisory boards and KOL primary research, to address key evidence uncertainties |
Table 1: Strategic priorities for Market Access leaders under the EU HTA framework
Act early, Win later: Building a Market Access advantage in the EU JCA era
Proactive preparation is essential in the new EU HTA era. Market access leaders who actively monitor and analyse early JCAs can gain a deeper understanding of how evidence is assessed, how Member State perspectives influence outcomes, and where discrepancies may still arise at the national level. By engaging early and strategically, market access leaders can effectively prepare for future launches, reduce risk, and maximise access outcomes.
Contact Remap Consulting by clicking here to discuss how we can assist you in navigating the JCA, JSC, and PICO strategy.
References
- European Commission. New EU rules on Health Technology Assessment open up a new era for patient access to innovation. European Commission,. Accessed 12/01/2026, https://ec.europa.eu/commission/presscorner/detail/en/ip_25_226
- European Commission. Joint Clinical Assessments. European Commission,. Accessed 12/01/2026, https://health.ec.europa.eu/health-technology-assessment/implementation-regulation-health-technology-assessment/joint-clinical-assessments_en?prefLang=de
- Member State Coordination Group on Health Technology Assessment (HTACG). Guidance on the scoping process. Member State Coordination Group on Health Technology Assessment (HTACG),. Accessed 12/01/2026, https://health.ec.europa.eu/document/download/7be11d76-9a78-426c-8e32-79d30a115a64_en?filename=hta_jca_scoping-process_en.pdf
- European Federation of Pharmaceutical Industries and Associations (EFPIA). From guidance to implementation: EFPIA’s reflections on EU HTA scoping & PICO exercises. European Federation of Pharmaceutical Industries and Associations (EFPIA),. Accessed 12/01/2026, https://www.efpia.eu/news-events/the-efpia-view/blog-articles/from-guidance-to-implementation-efpia-s-reflections-on-eu-hta-scoping-pico-exercises/
- Member State Coordination Group on Health Technology Assessment (HTACG). Annual Work Programme 2026. Member State Coordination Group on Health Technology Assessment (HTACG),. Accessed 12/01/2026, https://health.ec.europa.eu/document/download/81db097f-a9cb-4404-837c-115b4289321d_en?filename=hta_hatcg_2026-awp_en.pdf
- Institute for Quality and Efficiency in Health Care (IQWiG). First EU HTAs with IQWiG involvement launched. Institute for Quality and Efficiency in Health Care (IQWiG),. Accessed 12/01/2026, https://www.iqwig.de/en/presse/press-releases/press-releases-detailpage_158016.html?
- European Commission. Joint Scientific Consultations. European Commission,. Accessed 12/01/2026, https://health.ec.europa.eu/health-technology-assessment/implementation-regulation-health-technology-assessment/joint-scientific-consultations_en
- European Commission. Procedural Guidance for JCA medical devices and in vitro diagnostic medical devices. European Commission,. Accessed 12/01/2026, https://health.ec.europa.eu/publications/procedural-guidance-jca-medical-devices-and-vitro-diagnostic-medical-devices_en