Originally published 7th September 2023, and updated 13th September 2024
Introduction to orphan drug prices and market share
Orphan drugs are designated as they are used to treat very rare diseases with a small patient population or where the medicine is unlikely to generate sufficient profit to justify R&D costs. However, their introduction to the market is the key driver of the overall spending growth on drugs and sets significant financial challenges for healthcare systems. In 2023, the global market for orphan drugs was projected to reach $217 billion by 2024, but new forecasts suggest this number could exceed $415 billion by 2028, driven by a 12.2% Compound Annual Growth Rate (CAGR)¹². This unexpected growth is due to increased R&D investment, a rising prevalence of rare diseases, and favourable government policies promoting orphan drug development¹⁰,¹¹. As of 2024, orphan drugs are expected to account for nearly 20% of total prescription drug sales¹². However, significant pricing differences remain between the U.S. and Europe, which this article will explore.
What is the extent of the differences between the US and Europe?
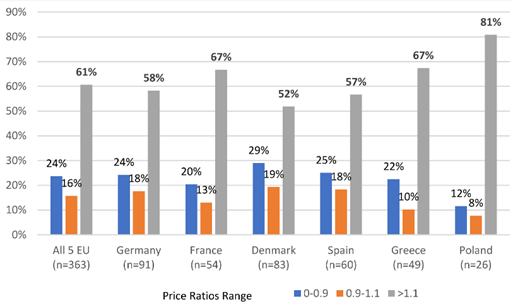
From launch until loss of orphan status, U.S. orphan drug prices continue to be significantly higher than in Europe. Recent analysis now shows U.S. prices are, on average, 1.7 times higher than in Europe¹² (increased from 1.64 times in September 2023). Additionally, the gap between U.S. and European orphan drug prices is widening. In 2024, the price ratio for multiple myeloma drug Pomalyst/Imnovid has increased further. As of 4th September 2024, the U.S. price is $27,500 compared to $12,000 in the UK, a ratio of 2.3¹², reflecting ongoing discrepancies in the pricing mechanisms between the two regions.
Is there a difference between single use and chronic treatments?
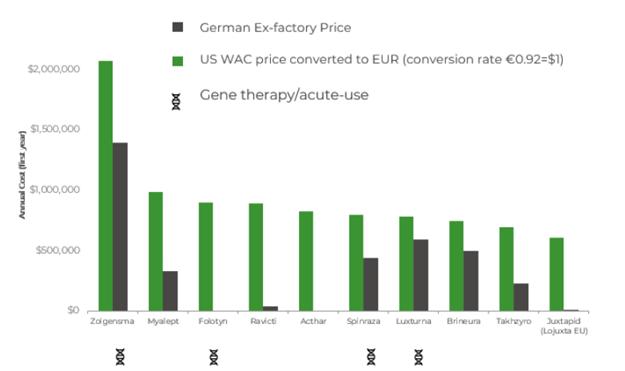
Acute treatments tend to have greater expense upfront compared to chronic treatments. However, as they are single-use and often curative, they can be considered good value over a patient’s lifetime. For example, Zolgensma, with a U.S. WAC price of $1.9 million, can be compared to chronic alternatives like Spinraza, which has a price point ten times lower than Zolgensma.
New data from 2024 highlights that single-use curative therapies, though expensive upfront, continue to offer long-term cost advantages compared to chronic treatments¹². Zolgensma, now priced at $2.1 million in the U.S. in 2024, continues to rival chronic alternatives like Spinraza in terms of lifetime cost¹². This has led to acute therapies, particularly gene therapies, being seen as cost-effective over a patient’s lifetime, which is a critical discussion point in global healthcare systems.
Why are orphan drug prices higher in the US compared to Europe?
When comparing the difference in price between the U.S. and Europe for orphan and non-orphan drugs, the relative differences are comparable, suggesting the reasons for higher U.S. prices are the same for both categories. The key driver is the lack of Health Technology Assessments (HTA) in the U.S., where pricing is driven by free-market negotiations between healthcare insurers and manufacturers.
In 2024, new regulatory discussions and initiatives are being explored to control orphan drug prices in the U.S.. The Institute for Clinical and Economic Review (ICER) has proposed expanding outcomes-based contracts and value-based pricing to reduce costs¹²,¹¹. In Europe, price negotiations continue to centre on the extent of additional benefit to patients, along with more aggressive cost-containment measures.
Conclusion
In conclusion, U.S. orphan drug prices remain substantially higher than in Europe, driven by the lack of a national health technology assessment (HTA) framework in the U.S. and the strong negotiating power of European systems. While ongoing reforms like the ICER proposals and the Inflation Reduction Act (IRA) have sparked discussions on price reductions, little progress has been made so far¹². The situation is unlikely to change until the scope of these reforms expands, but discussions around innovative pricing models are expected to gain momentum through 2025. For more information orphan medicine market access in Europe and the growing challenges ahead in our upcoming webinar on the 28th September, find out more about this on our website.
Sources:
1. Research and Markets. Global Orphan Drug Report 2020-2024: Over 7,600 Orphan Drug Designations Covered Across the $217 Billion Market. Accessed 03/09/2023, https://www.prnewswire.com/news-releases/global-orphan-drug-report-2020-2024-over-7-600-orphan-drug-designations-covered-across-the-217-billion-market-301369022.html
2. Villa F, Filippo A, Pierantozzi A, et al. Orphan Drug Prices and Epidemiology of Rare Diseases: A Cross-Sectional Study in Italy in the Years 2014–2019. Frontiers in Medicine. 2022;9doi:10.3389/fmed.2022.820757
3. Zelewski P, Wojna M, Sygit K, Cipora E, Gaska I. Comparison of US and EU Prices for Orphan Drugs in the Perspective of the Considered US Orphan Drugs Act Modifications and Discussed Price-Regulation Mechanisms Adjustments in US and European Union. International Journal of Environmental Research and Public Health. 2022;19(19)doi:10.3390/ijerph191912098
4. Micromedex. Redbook. Accessed 04/09/2023, https://www.micromedexsolutions.com/micromedex2/librarian/CS/E8DB86/ND_PR/evidencexpert/ND_P/evidencexpert/DUPLICATIONSHIELDSYNC/3B1933/ND_PG/evidencexpert/ND_B/evidencexpert/ND_AppProduct/evidencexpert/ND_T/evidencexpert/PFActionId/redbook.FindRedBook?navitem=topRedBook&isToolPage=true
5. National Institute of Health and Care Excellence. British National Formulary. Accessed 04/09/23, https://bnf.nice.org.uk/
6. Kang S-Y, DiStefano M, Socal M, Anderson G. Using external reference pricing In medicare part D to reduce drug price differentials with other countries. Pharmaceuticals and Medical Technology. 2019;38(5)doi:10.1377/hlthaff.2018.05207
7. Remap Consulting. Are acute therapies and curative drugs more affordable than chronic treatments in rare diseases? Accessed 04/09/2023, https://remapconsulting.com/orphan-drugs/are-acute-therapies-and-curative-drugs-more-affordable-than-chronic-treatments-in-rare-diseases/
8. Koyuncu A, Herold S. Inside EU Life Sciences: Updates on Legal Developments in the EU Life Sciences Industry Accessed 06/09/2023, https://www.insideeulifesciences.com/2022/10/26/germany-significantly-tightens-drug-pricing-and-reimbursement-laws/
9. Onakpoya I, Spencer E, Thompson M, Heneghan C. Effectiveness, safety and costs of orphan drugs: an evidence-based review. BMJ Open. 2015;5(6)doi:10.1136/bmjopen-2014-007199
10. Mitchell G. Zynteglo highlights stark differences between EU and US markets Accessed 06/09/2023, https://www.lcp.com/our-viewpoint/2022/10/zynteglo-highlights-stark-differences-between-eu-and-us-markets/
11. Institute for Economic Review. The Next Generation of Rare Disease Drug Policy: Ensuring Both Innovation and Affordability 2022. https://icer.org/wp-content/uploads/2022/04/ICER-White-Paper_The-Next-Generation-of-Rare-Disease-Drug-Policy_040722.pdf
12. Chambers J, Clifford K, Enright D, Neumann P. Follow-On Indications for Orphan Drugs Related to the Inflation Reduction Act. Health Policy. 2023;6(8)doi:10.1001/jamanetworkopen.2023.29006