While missing an EU Joint Scientific Consultation (JSC) is suboptimal, the greater strategic concern is whether it disrupts progress on the wider evidence plan for EU market access.¹ Where decisions on comparator, endpoints, or population are deferred pending JSC input, reimbursement strategy can become unnecessarily constrained by process timing rather than anchored in evidence and launch objectives. With the HTA Coordination Group planning only 8 to 12 Joint Scientific Consultations (JSCs) for medicinal products and 2 to 5 for medical devices, securing a slot remains uncertain.²
The good news is that a formal JSC slot is not the only route to pressure testing evidence strategies. Targeted national HTA scientific advice, combined with well-structured advisory boards, can also generate robust insights: testing the credibility of comparators and endpoints, anticipating likely assessor concerns, and translating uncertainty into clear evidence priorities while there is still scope to strengthen the evidence package. ³⁻⁵
This section outlines how JSC-level insight can still be achieved in the absence of a formal JSC opportunity.
What JSC really gives you (and what it does not)
A JSC can provide early, structured challenge to the evidence plan. Its stated purpose is to help health technology developers understand the information, data, analyses, and other evidence likely to be needed for a subsequent Joint Clinical Assessment (JCA), with discussion focused on key clinical design choices such as comparator, interventions, outcomes, and patient populations.6
The value of JSC lies in both the chance to recalibrate your evidence strategy ahead of submission to ensure alignment with JSC requirements, and to adjust whilst there is still time. What makes JSC distinct is that the challenge comes directly from coordinated, multi-country assessors.
It also offers a broader HTA perspective than a single national advice process. For medicinal products, the process is run through the HTACG JSC subgroup, with an assessor and co-assessor, and it includes input from patients, clinical experts, and other relevant experts through the consultation process and discussion meeting.7 Moreover, a JSC can be co-ordinated in parallel with EMA scientific advice, which can reduce duplication and help align regulatory and HTA evidence discussions more efficiently.7
When JSC timing or process burden pose as significant constraints, manufacturers may consider alternative routes to pressure-test comparators, endpoints, and surfacing likely HTA challenges. On many occasions, seeking national scientific advice in key markets and complementing it with well-designed EU-advisory boards can provide a robust approach to inform both EU-level and national reimbursement strategies.
Table 1. How the JSC compares to alternatives
| Considerations | JSC | National HTA scientific advice | Advisory boards |
|---|---|---|---|
| Obtain multi-country HTA perspectives (PICOs) | +++ | – | ++ |
| Can be integrated with EMA scientific advice and reduce duplication | +++ | – | – |
| Captures targeted national-level requirements and translates them into local reimbursement implications | – | ++ (single country focus) | +++ (multi-country focus) |
| Feasibility | + (higher resource use and process burden) | ++ | +++ (lower resource use and process burden) |
| Ease of access | Limited slots | More predictable | Highly flexible |
What should manufacturers do now to secure alignment with JCA and national-level evidence requirements?
Where a JSC is not feasible or strategically deprioritised, evidence planning should still be anchored in external expert input. The decision is therefore not whether advice is needed, but which route will generate the most actionable outputs at the right point in the development process.
In practice, this means seeking early national HTA scientific advice in the markets most likely to shape the JCA, complementing this with a multi-country advisory board to test the evidence package at a more granular level, and translating that feedback into a concrete evidence-optimisation strategy (Figure 1).
Figure 1. Sequence of steps for scientific advice
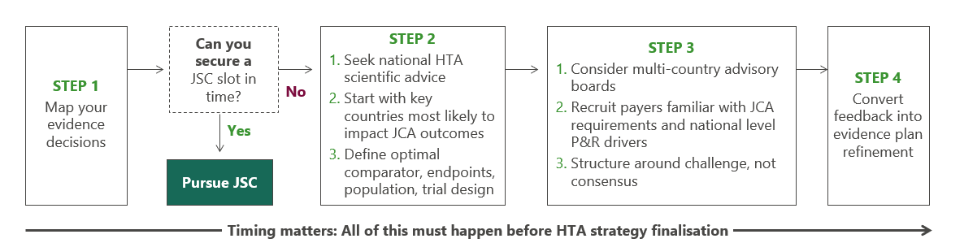
Although the JSC is a valuable mechanism, current capacity constraints are structural, and the resources needed for preparation and process navigation should not be underestimated. More importantly, allowing limited JSC slot availability to delay critical evidence decisions may shift the risk to a later stage, when course correction may be more difficult.²
Critically, multi-country advisory boards and early national-level scientific advice ensure that evidence decisions are incorporated before Phase 3 protocol finalisation. That’s the window where scientific advice has most impact and can still be translated into real tactical actions. Once the Phase 3 protocol is locked, the options narrow significantly, and any gaps in the evidence package become much harder, and more expensive, to address.
What next?
When planning for scientific advisory boards, it is important to ensure they are designed to generate actionable insights rather than simply validating an existing plan or reinforcing challenges.
The approach that tends to make these strategic meetings strongest are:
- Ensure a sharply defined focus: The most valuable advisory boards are built around specific, well-defined evidence questions rather than very broad or untargeted discussions.
- Ensure an expert mix that reflects the real decision environment: Advisory boards are stronger when participants are selected for the expertise needed to interrogate the evidence package meaningfully, not for cost or convenience.
- Leverage virtual and hybrid formats to widen participation and optimise inclusion of the right experts to match the specific evidence development questions.
Remap brings long-standing experience in designing and delivering EU and global advisory boards that help clients challenge assumptions, refine evidence strategies, and strengthen decision-making at critical points in development. Contact us to find out more about our unique approach.
Abbreviations: EU: European Union; HTA: Health Technology Assessment; JCA: Joint Clinical Assessment; JSC: Joint Scientific Consultation; RWE: Real-world evidence; SAP: Statistical analysis plan
Download: JSC Alternatives Reimbursement Risk Checklist
A practical checklist for teams facing Joint Scientific Consultation timing or capacity constraints. It helps assess comparator, endpoint and evidence plan exposure — and highlights where early national HTA advice or advisory boards can reduce reimbursement risk before pivotal decisions are finalised.
References
- European Commission. Joint scientific consultations [Internet]. Brussels: European Commission; [cited 2026 Mar 13]. Available from: https://health.ec.europa.eu/health-technology-assessment/implementation-regulation-health-technology-assessment/joint-scientific-consultations_en
- European Commission. HTACG annual work programme 2026 [Internet]. Brussels: European Commission; 2025 [cited 2026 Apr 1]. Available from: https://health.ec.europa.eu/document/download/81db097f-a9cb-4404-837c-115b4289321d_en?filename=hta_hatcg_2026-awp_en.pdf&prefLang=it
- Haute Autorité de Santé. Early dialogues [Internet]. Saint-Denis La Plaine: HAS; [cited 2026 Mar 13]. Available from: https://www.has-sante.fr/jcms/c_1625763/en/early-dialogues
- National Institute for Health and Care Excellence. NICE technology appraisal and highly specialised technologies guidance: the manual [Internet]. London: NICE; 2022 Jan 31 [updated 2025 Dec 17; cited 2026 Mar 13]. Available from: https://www.nice.org.uk/process/pmg36/chapter/evidence-2
- European Medicines Agency. Parallel scientific advice and special development aspects or product types [Internet]. Amsterdam: EMA; [cited 2026 Mar 13]. Available from: https://www.ema.europa.eu/en/human-regulatory-overview/research-development/scientific-advice-protocol-assistance/parallel-scientific-advice-special-development-aspects-or-product-types
- European Parliament, Council of the European Union. Regulation (EU) 2021/2282 of the European Parliament and of the Council of 15 December 2021 on health technology assessment and amending Directive 2011/24/EU [Internet]. Off J Eur Union. 2021 [cited 2026 Mar 13]. Available from: https://eur-lex.europa.eu/eli/reg/2021/2282/oj/eng
- HTA Coordination Group. Procedural guidance for joint scientific consultations (JSC) on medicinal products (MP) [Internet]. Version 5.5. Brussels: European Commission; 2024 [cited 2026 Apr 1]. Available from: https://health.ec.europa.eu/document/download/bbf25ce0-81eb-4c2a-b09a-e08d4611c165_en?filename=hta_jsc_mp_procedural-guidance_en.pdf